Lime 
- The word “lime” refers to products derived from heating limestone.
- It originates with its earliest use as building mortar and has the sense of “sticking or adhering“
- The rocks and minerals from which these materials are derived, typically limestone or chalk, are composed primarily of calcium carbonate (CaCO3).
Quick Lime (CaO)
Pure lime, generally called quick lime, is a white oxide of calcium. Much of commercial quick lime, however, contains more or less magnesium oxide, which gives the product a brownish or grayish tinge. Quick lime is obtained after the calcination of limestone. It is also called caustic lime. It is capable of slaking with water and has no affinity for carbonic acid. The specific gravity of pure lime is about 3.40.
(Calcination is used to mean a thermal treatment process in the absence or limited supply of air or oxygen applied to ores and other solid materials to bring about a thermal decomposition. A calciner is a steel cylinder that rotates inside a heated furnace and performs indirect high-temperature processing (550-1150 °C, or 1000-2100 °F) within a controlled atmosphere.)
Fat Lime
has high calcium oxide component and, sets and hardens by the absorption of CO2 from atmosphere. These are manufactured by burning marble, white chalk, calcareous tufa, pure limestone, sea shell and coral.
Hydraulic Lime
contains small quantities of silica, alumina, iron oxide in chemical combination with calcium oxide component. These are produced from carboniferous limestones and magnesian limestone. It has the property to set and harden under water.
Hydrated Lime
When quick lime is finely crushed, slaked with a minimum amount of water, and screened or ground to form a fine homogeneous powder the product is called hydrated lime.
Lump Lime
is the quick-lime coming out of the kilns.
Milk Lime
is a thin pourable solution of slaked lime in water.
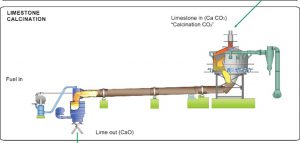
Lime Production
When limestone (calcium carbonate) is heated, at about 1000 °C it undergoes thermal decomposition. It loses carbon dioxide and turns into quicklime (calcium oxide).
The reaction is carried out in specially constructed lime kilns(a kiln is a high temperature oven). Limestone is added at the top and quicklime is removed from the bottom in a continuous process.
Lime Cycle
Lime Production
Uses of Lime
- Lime has been used in building techniques for over 5,000 years. Archaeological evidence shows it to have been in existence for this time frame due to its resilience, durability, and water resistant qualities.
- The Romans used lime extensively in their building program in Britain, and refined its application into mortars and plasters, which remained the principal surface finish for buildings until the nineteenth century, when cements took over this function. For this reason, many historic buildings in the UK contain large amounts of lime within their fabric
- Also used for pointing and plastering.
Lime vs Cement
Impurities in Limestone
(Click to see the Impurities in Limestone)
Due to the great diversity in the usage of buildings and installations and the various processes of production, a great variety of requirements are placed upon building materials calling for a very wide range of their properties: strength at low and high temperatures, resistance to ordinary water and sea water, acids and alkalis etc. Also, materials for interior decoration of residential and public buildings, gardens and parks, etc. should be, by their very purpose, pleasant to the eye, durable and strong. Due to the great diversity in the usage of buildings and installations and the various processes of production, a great variety of requirements are placed upon building materials calling for a very wide range of their properties: strength at low and high temperatures, resistance to ordinary water and sea water, acids and alkalis etc. Also, materials for interior decoration of residential and public buildings, gardens and parks, etc. should be, by their very purpose, pleasant to the eye, durable and strong. Due to the great diversity in the usage of buildings and installations and the various processes of production, a great variety of requirements are placed upon building materials calling for a very wide range of their properties: strength at low and high temperatures, resistance to ordinary water and sea water, acids and alkalis etc. Also, materials for interior decoration of residential and public buildings, gardens and parks, etc. should be, by their very purpose, pleasant to the eye, durable and strong. Due to the great diversity in the usage of buildings and installations and the various processes of production, a great variety of requirements are placed upon building materials calling for a very wide range of their properties: strength at low and high temperatures, resistance to ordinary water and sea water, acids and alkalis etc. Also, materials for interior decoration of residential and public buildings, gardens and parks, etc. should be, by their very purpose, pleasant to the eye, durable and strong.